
New Mobile Room UVC Brochures with How to Estimate UVC Disinfection Dosage
We are frequently asked how we estimate the UVC dosage needed to reduce microorganisms in the environment. Scientists to date have established so called "k-values," which are disassociation constants that determine how much UVC energy will need to be applied to break the DNA or RNA bonds in viruses, bacteria or fungal spores. The microorganism will no longer be able to replicate; hence, is no longer capable of causing disease.
The tables on this page show estimated UVC dose (mJ/cm2) values and cycle times can be estimated:
Learn more about the dosage values for each device (below).
MRS14-4
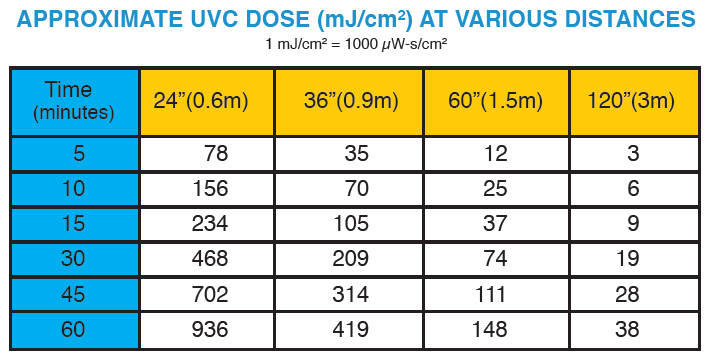
MRS 33-8
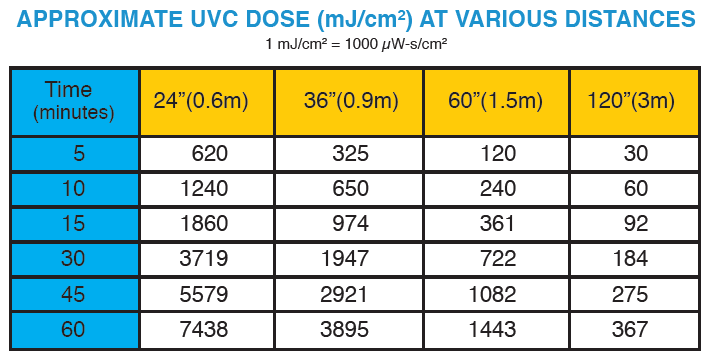
MRS 45-12
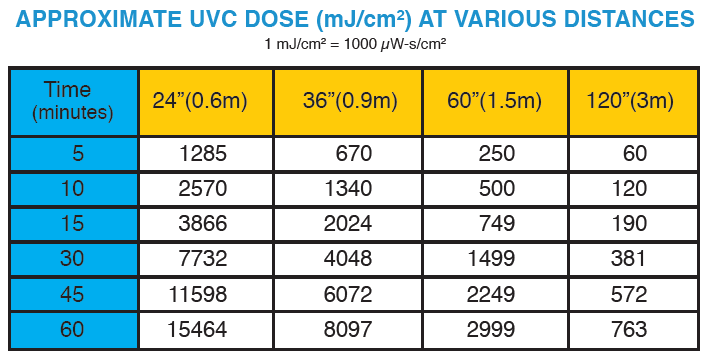
Visit the Disinfection Update E-newsletter archives (which begin with March 2020) to read helpful stories about the effectiveness of UVC Disinfection.
None of the American Ultraviolet UVC products detailed above are certified, or approved under any applicable laws, as a medical device, and as such, American Ultraviolet, and its Representatives and Distributors, do not currently intend for them to be used as medical devices anywhere globally. Products have not been evaluated by the FDA.
