In-Duct DC Series
American Ultraviolet High-output DC Series HVAC UVC germicidal fixtures can be mounted in various configurations for optimum pass-by air decontamination and/or to irradiate cooling coils and drain pans. Lamps and electronics can be accessed from outside the duct or plenum. "Green" lamps (contain < 8mg of mercury) extend inside to span supply ducts and/or cooling coils.
DC Series HVAC air duct system fixtures are offered in two and four-lamp configurations that can be mounted singly, in built-up banks, or in parallel configurations in a variety of locations, including coils, drain pans, ductwork, mixed air plenums and exhaust systems. The DC series conforms to UL Std 1598 and is certified to CSA Std C22.2#250.
In-Duct Intensity Control Rack (ICR) Series
High-output ICR Series fixtures from American Ultraviolet are designed specifically for healthcare airstream disinfection applications. They are ideal for large air handling systems in commercial, industrial, healthcare, and institutional buildings. These ready to install framed structures can be easily mounted in duct systems and on coils to provide the desired airstream disinfection from unwanted microorganisms travelling in the airstream.
ICR Series fixtures not only decontaminate air, but they also use UVC energy to remove biofilms from cooling coils and drain pans. The High-output ICR Series can be mounted within minutes to plenum walls, or to frame assemblies that span supply ducts and/or cooling coils. These fixtures are offered in various lamp configurations and come standard in five different widths (18”, 24”, 36”, 48”, & 60”). Frames can be mounted side by side or stacked vertically to cover the entire plenum or coil.
Benefits
- Reduce bacteria, viruses and fungi with one pass through ICR
- Each system is custom sized to individual duct and desired microbial reduction rate at no additional charge
- Installs in minutes; comes with ready assembled frame structure that easily attaches to plenum wall
- Installation down-stream offers removal or prevention of biofilm build-ups on cooling coil; and restores coil back to optimum heat transfer properties
- “Green” lamps contain ≤ 8mg of Mercury
- Exclusive 2-year (17,000 hours) guarantee on high output lamp life
- No maintenance required, other than changing lamps every 2 years
To learn more about ICR Series fixtures, including technical specifications, please visit our technical library where you can download and print literature.

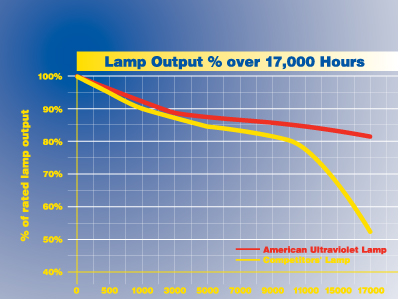
Prolonged, direct exposure to UVC light can cause temporary skin redness and eye irritation. American Ultraviolet systems are designed with safety in mind and, when properly installed by a professional contractor, do not allow exposure to UV irradiation and allow for safe operation and maintenance.

Example of In-Duct Disinfection
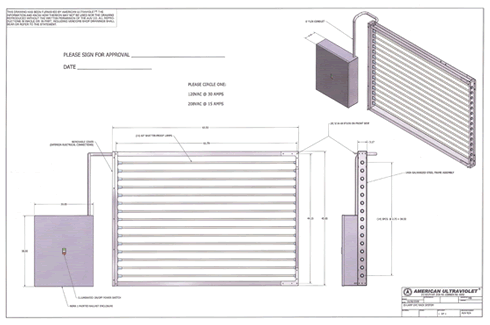
Specifications
- Each assembly includes fully wired and enclosed power supply, electronic ballast(s), frame assembly, lamp holders, and high output lamp(s)
- “Moisture-Proof” construction incorporates a moisture proof wire lead connector assembly
- High output (800ma), green lamps (≤ 8mg Mercury)
- Lamps produce no ozone or other secondary contaminants
Prolonged, direct exposure to UVC light can cause temporary skin redness and eye irritation. American Ultraviolet systems are designed with safety in mind and, when properly installed by a professional contractor, do not allow exposure to UV irradiation and allow for safe operation and maintenance.
None of the American Ultraviolet UVC products detailed above are certified, or approved under any applicable laws, as a medical device, and as such, American Ultraviolet, and its Representatives and Distributors, do not currently intend for them to be used as medical devices anywhere globally. Products have not been evaluated by the FDA.
